11.05.2026
3 minutes of reading
The emergence of complex pollutants such as per- and polyfluoroalkyl substances (PFAS) in natural environments poses a major environmental and health challenge, and the associated new regulations require appropriate treatment methods. To this end, adsorption phenomena at the solid-liquid interface are critical to the development of rapid and selective filtration methods. That is because many pollution control processes involve a filtration step based on the adsorption of pollutants on porous grains, hence the need for a better understanding of adsorption phenomena.
Beyond traditional models and semi-empirical laws
The amphiphilic nature of PFAS (i.e., compounds with both hydrophobic and hydrophilic parts) causes them to behave in complex ways when they are in solution and/or at a solid-liquid interface. In particular, aggregate formation can be observed when their concentration exceeds the critical micelle concentration (CMC) [1]. The resulting intermolecular interactions between adsorbed molecules significantly alter the adsorption isotherms1 and kinetics. Consequently, conventional models (e.g., Henry or Langmuir models2) that neglect these lateral interactions fail to account for the cooperative effects generated between molecules, particularly when the isotherms exhibit inflection points3 [1,2]. Similarly, experimental adsorption kinetics often deviate from classical first-order laws, where the time evolution of the adsorbed amount is proportional to the deviation from the equilibrium adsorbed amount. It is for this reason that (semi-)empirical laws are frequently used to describe the experimental data obtained, without any knowledge of, or consideration for, the underlying adsorption mechanisms.
To address this issue, a PhD research project [3] proposes modeling of the thermodynamic and kinetic aspects of adsorption based on three key mechanisms:
- reservoir depletion (associated with a change in the concentration of pollutants in solution in the medium), which is significant in diluted systems,
- surface saturation (associated with the adsorbent's limited adsorption capacity)
- and lateral interactions between adsorbates [3].
Development of an adsorption model
Firstly, a thermodynamic model incorporating two- and three-molecule interactions was developed [Figure 1] in order to describe all the adsorption isotherms observed for amphiphilic molecules such as PFAS, as well as surfactants and certain antibiotics (L, S, and LS curves exhibiting 0, 1, or 2 changes in convexity). The coupling between depletion and saturation, in turn, leads to mixed-order kinetics4, which explains the deviations observed experimentally compared to first-order kinetics. Finally, the effects of cooperative interactions on kinetic parameters are accounted for through adsorption and desorption constants (which depend on the amount adsorbed), affecting both the final equilibrium state and the time required to reach it.
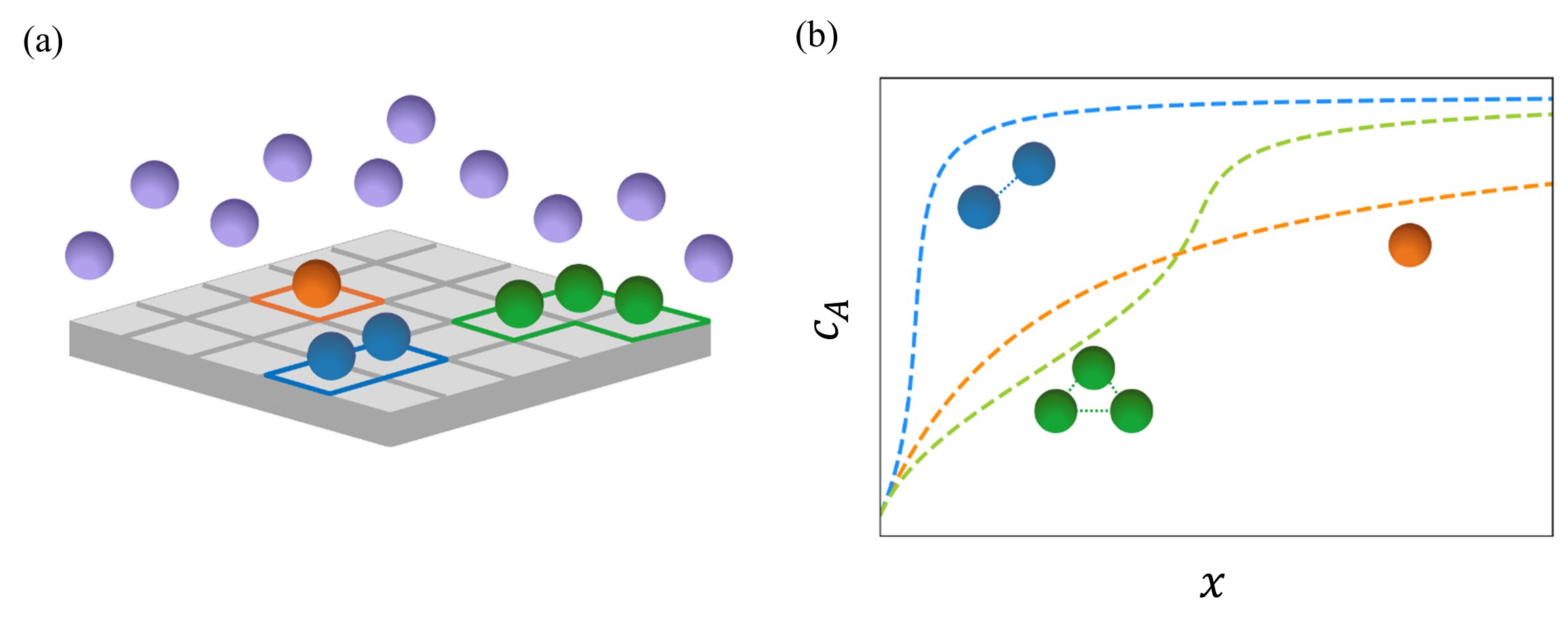
Extrapolation to highly diluted media
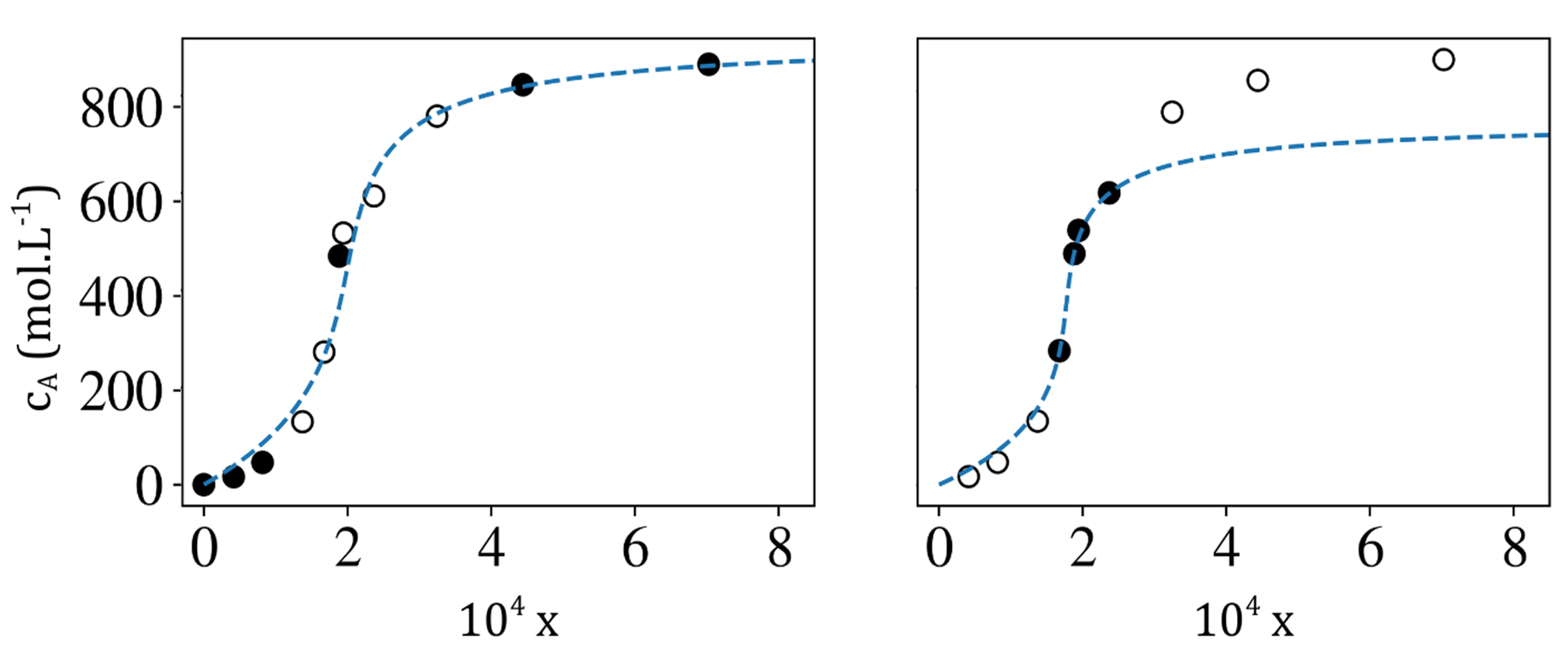
This adsorption model not only allows for a better interpretation of experimental measurements but also enables the extrapolation of results obtained at high concentrations to highly diluted conditions [Figure 2]. These new capabilities are particularly important for water treatment in natural environments, where pollutant concentrations are limited to a few ng/L, whereas with current technologies, it is extremely difficult to measure adsorption below a few µg/L [4].
Perspectives : évaluation et optimisation des filtres d’adsorption
En perspective, ce modèle pourrait être implémenté dans des simulations de type Lattice Boltzmann (méthode numérique pour la résolution d’équations de transport) afin de quantifier l’impact sur le transport des adsorbats des isothermes spécifiques à un couple d’adsorbat-adsorbant. A terme, cette avancée permettrait d’évaluer la performance de filtres basés sur l’adsorption et d’optimiser leur utilisation pour des composés spécifiques tels que les PFAS.
1 In thermodynamics, a liquid-phase adsorption isotherm is the relationship, at constant temperature, between the adsorbed concentration cA and the concentration of the adsorbate in the solution at equilibrium ce. It should be noted that the adsorbed concentration cA [in moles of adsorbate per unit volume of solid] is defined as the product of the adsorbed amount [in moles of adsorbate per unit mass of solid] and the density of the adsorbent solid [mass per unit volume of solid]
2 In Henry's model, the adsorbed concentration is proportional to the solution concentration (linear relationship, where cA ~ ce), whereas the Langmuir model accounts for surface saturation, thus converging to a plateau (L-shaped curve).
3 This change in convexity indicates an adsorption mechanism dominated by the formation of surface aggregates.
4 i.e., the time variation of the adsorbed concentration cA(t) is a function of cA(t) and cA(t)2
References:
[1] Johnson, R. L., Anschutz, A. J., Smolen, J. M., Simcik, M. F., & Penn, R. L. (2007). The adsorption of perfluorooctane sulfonate onto sand, clay, and iron oxide surfaces. Journal of Chemical & Engineering Data, 52(4), 1165-1170.
>> DOI : https://doi.org/10.1021/je060285g
[2] Soares, S. F., Rocha, M. J., Ferro, M., Amorim, C. O., Amaral, J. S., Trindade, T., & Daniel-da-Silva, A. L. (2019). Magnetic nanosorbents with siliceous hybrid shells of alginic acid and carrageenan for removal of ciprofloxacin. International journal of biological macromolecules, 139, 827-841.
>> DOI : https://doi.org/10.1016/j.ijbiomac.2019.08.030
[3] Ben Amor, N., Bauer, D., Braconnier, B., & Coasne, B. (2025). Adsorption Thermodynamics and Kinetics of Simple and Complex Fluids: Physical Modeling of Surface Saturation, Reservoir Depletion, Lateral Interactions, and Collective Effects. Langmuir, 42(1), 1348-1357.
>> DOI : https://doi.org/10.1021/acs.langmuir.5c05404
[4] Ben Amor, N., Braconnier, B., Coasne, B., Nieto-Draghi C., & Bauer, D. (2026). Reconstructing Adsorption Isotherms for Complex Molecules from Limited Data: A New Method Combining Extrapolation and Physics-based Modeling. Presented at the 2nd French and German conference on Adsorption. Article currently under Peer Review in Advanced Theory and Simulation.
Scientific contact: Daniela Bauer





