15.12.2021
2 minutes of reading
 Electrolyte Thermodynamics (EleTher) is known to be one of the key challenges for developing many industrial processes. To take up this challenge, the EleTher JIP aimed at creating a community of practice on electrolytes modeling. After three years of collaboration and research, significant progress has been made on the understanding of fluids with electrolytic species.
Electrolyte Thermodynamics (EleTher) is known to be one of the key challenges for developing many industrial processes. To take up this challenge, the EleTher JIP aimed at creating a community of practice on electrolytes modeling. After three years of collaboration and research, significant progress has been made on the understanding of fluids with electrolytic species.
Overall context: fluid mixtures containing ions need investigation
Many applications in the process industry deal with fluids that contain electrolytic species. However, while thermodynamic models for neutral molecules are now well established, there are still many unanswered issues related to the presence of ions in a fluid mixture.
- There is no agreement in the scientific community about the best model to use for combined long range and short-range interactions. The industrial models used to date often have a very large number of parameters.
- The presence of chemical equilibrium implies the need to work with reactive algorithms searching for a global minimum (phase and chemical).
- Several phases may coexist (several liquids in addition to vapor and several solids). This implies a rigorous phase stability analysis.
- Considering the complexity of the industrial systems, the data are often insufficient, meaning that data extrapolation methods may be needed.
A three-step process: data collection, data extrapolation, model parametrization with sensitivity analysis
To address these issues, seven industrial companies have joined a Joint Industry Project (JIP), called EleTher, launched in 2019 by IFPEN for three years. In addition to the technical work, the consortium aimed at creating an industrial community promoting research on this topic.
a Joint Industry Project (JIP), called EleTher, launched in 2019 by IFPEN for three years. In addition to the technical work, the consortium aimed at creating an industrial community promoting research on this topic.
The first step of this dissemination work has been the building of the EleTher website. Other actions included an active involvement in the Industrial Use of Thermodynamics (IUT) symposium held at the ESAT 2021 conference, a contribution to a ‘Spotlight Talk’ webinar (“Electrolyte Thermodynamics challenges from industrial needs to academic research” - watch replay) organized by the European Federation of Chemical Engineering (EFCE), and the publication of a scientific paper in the Journal of Chemical & Engineering Data (“Data Analysis for Electrolyte Systems: A method illustrated on alkali halides with water”).
The technical work focused on the development of a workflow in three steps:
- Data collection: data are essential for the model development, but they may be inconsistent or outside of the range of industrial interest. A consistency analysis is therefore essential.
- Extrapolation of the existing data to the industrial operating conditions: trends that can be identified in the data must be extrapolated, generally well beyond reasonable limits. This requires educated guesses that are based on an understanding of the physical phenomena involved. Advanced molecular models can be used for this purpose, but the limits of these tools must be respected.
- Once the true behavior of the fluid under the operating conditions is known, an industrial model must be parameterized before its introduction in a process simulator. This can become a real challenge when there are no clear guidelines, as these models may contain many such parameters. When too many parameters are fitted on too little data, the problem becomes overdetermined and impossible to use. A sensitivity analysis may help to identify the parameters that are most important.
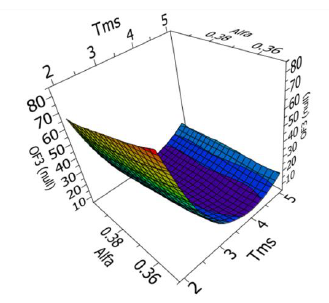
The investigation of vapor-liquid equilibria on reactive systems have demonstrated that the presence of new species in solution in the liquid phase significantly modifies the phase behavior. An example is given hereafter concerning the addition of NaOH on the bubble pressure of a solution (water + acetic acid + ethanol).
.jpg)
Points: total dissociation.
Solid lines: partial dissociation (calculated with data from different sources)
Further work to come in a follow-up JIP
From the current results, it can be concluded that the correct resolution of the problem often requires additional information (speciation).
Further work along these lines is planned, both from a research perspective in the EleTher Chair, and in a follow-up JIP.
Meanwhile, the results of the JIP’s research will be useful in several industrial fields. For instance, they can be implemented for the understanding of phenomena occurring in various contexts: the processing of biomass, hydrometallurgical processes for metal recycling, or CO2 capture and storage.
>> Find out more: www.elether.fr








