26.03.2019
5 minutes of reading
To limit global warming and meet with commitments of the Paris Agreement (COP21), the reduction of greenhouse gas emissions resulting from human activities is a major objective.
To achieve this, CCS (Carbon Capture and Storage), consisting in storing CO2 underground, has been the focus of research over the past few years, along with other options such as CCU (Carbon Capture and Utilization), which gives the operation economic added value. Whatever the case, it is important to have a thorough understanding of the gas mixture under consideration, in such a way as to anticipate its interactions with the environments it may subsequently come into contact with.
For CO2 storage in various underground layers, IFPEN has taken part in the ANR Gaz Annexes (Co-injected Gases) and, more recently, SIGARRR (Simulations of the impact of co-injected gases on rock-reservoir reactivity) projects. These two projects were dedicated to the experimental and numerical study of the geological sequestration phase, within the framework of a security-focused approach (storage site integrity). Given that the injected gas contains impurities (NOx, SOx, H2S, N2, O2, etc.), they focused, in particular, on determining the impact of the latter on the physical properties of this gas, as well as its reactivity with water and/or minerals forming the reservoir and cap rocks.
Traditionally, the numerical simulations required by studies of this type use geochemical modeling based on empirical thermodynamic laws (activity models, equations of state, etc.), the parameters of which are calibrated using experimental data. In some cases, as a function of the compounds under consideration (toxicity, stability, etc.) and the targeted temperature and pressure conditions, the acquisition of new experimental data can prove extremely costly, hazardous or even impossible.
In recent years, in line with digitalization priorities, IFPEN has developed and used a range of molecular simulation techniques with a view to generating so-called “pseudo-experimental” data, for operating conditions that are difficult to achieve through experimentation [1-4].
The research carried out has proved successful since an excellent correlation between available experimental data and “pseudo-experimental” data has been obtained. The quality of these numerical forecasts concerns both properties (viscosity, density) and phase equilibrium diagrams (pressure-composition as presented in the figure below). No recalibration on the basis of experimental data is required since the calculations are intrinsically predictive for multicomponent systems. Thus it is a convenient method of generating data, particularly those used to optimize equations of state parameters that support geochemical models.
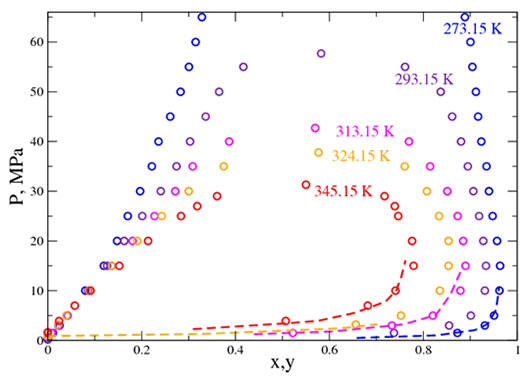
The x-axis represents the nitric oxide fraction. The dotted lines represent
the few reference experimental data available.
Scientific contact: benoit.creton@ifpen.fr
Publications
- Creton, B.; Lachet, V. Thermodynamic study of binary systems containing carbon dioxide and associated gases using molecular simulation techniques.Energy Research Network Conferences & Meetings, 2018. Presentation at the Congrès GHGT14.
>> https://ssrn.com/abstract=3365730 ?
- Creton, B.; Nieto-Draghi, C.; de Bruin, T.; Lachet, V.; El Ahmar, E.; Valtz, A.; Coquelet, C.; Lasala, S.; Privat, R.; Jaubert, J.-N. Thermodynamic study of binary systems containing sulphur dioxide and nitric oxide: measurements and modeling. Fluid Phase Equilibria, 2018, Vol. 461, p. 84-100.
>> DOI: 10.1016/j.fluid.2017.12.036
- Creton, B.; de Bruin, T.; Le Roux, D.; Duchet-Suchaux, P.; Lachet, V. Impact of associated gases on equilibrium and transport properties of a CO2 stream: Molecular simulation and experimental studies. International Journal of Thermophysics, 2014, Vol. 35, p. 256-276.
>> DOI: 10.1007/s10765-014-1592-6
- Lachet, V.; Creton, B.; de Bruin, T.; Bourasseau, E.; Desbiens, N.; Wilhelmsen, Ø.; Hammer, M. Equilibrium and transport properties of CO2+N2O and CO2+NO mixtures: Molecular simulation and equation of state modelling study. Fluid Phase Equilibria, 2012, Vol. 322-323, p. 66-78.
>> DOI: 10.1016/j.fluid.2012.03.011





