05.04.2024
3 minutes of reading
In the 21st century, the challenge of the ecological transition is requiring the chemicals industry to develop innovative processes in line with environmental, energy and societal constraints. There is a dual challenge here:
- new conversions need to be developed using renewable resources, the reactivity of which has yet to be fully mastered on a large scale;
- researchers need to design and propose innovations to make these conversions more efficient and less energy intensive.
The second aspect concerns processes and requires researchers to consider various avenues for progress: new reactors, reduction in the number of steps, combination of catalysts, etc.
Conversion of living organisms via biological catalysis
Starting from the principle that nature always uses energy efficiently and parsimoniously, biological catalysis, particularly enzymatic, is one of the solutions being studied. It is found in all living world conversion cycles and is particularly appropriate for converting natural renewable resources, such as sugars from biomass, with a very high selectivity, in aqueous media and at mild temperatures of between 20 and 40°C.
“Hybrid” catalysis reduces the number of steps
In order to reduce the number of intermediate separation and purification steps, which are energy and solvent intensive, so-called “hybrid” catalysis has emerged in the last decade consisting in combining biological catalysis and chemical catalysis. IFPEN’s researchers were pioneers in the field [1] in partnership with Lille University1 and co-published an exhaustive reference review on the subject [2].
Several hybrid approaches are possible to harness the advantages of both biological and chemical catalysis: the most economical consists in using the two types of catalyst simultaneously within the same reaction chamber in order to generate a chain of reactions. This approach requires a high degree of chemical and thermal compatibility between the two catalytic systems.
1 UCCS (Solids catalysis and chemistry unit) and Charles Violette Institute
A favored target for hybrid catalysis: glucose
A reaction of specific interest for hybrid catalysis is the conversion of glucose, an inexpensive, renewable natural resource that is found in abundance, into 5-hydroxymethylfurfural (HMF), a high added-value platform molecule for the fine chemicals sector (flavors, fragrances, resins, lubricants). Glucose is first converted into fructose, which is then dehydrated to form HMF (Fig. 1) [3].
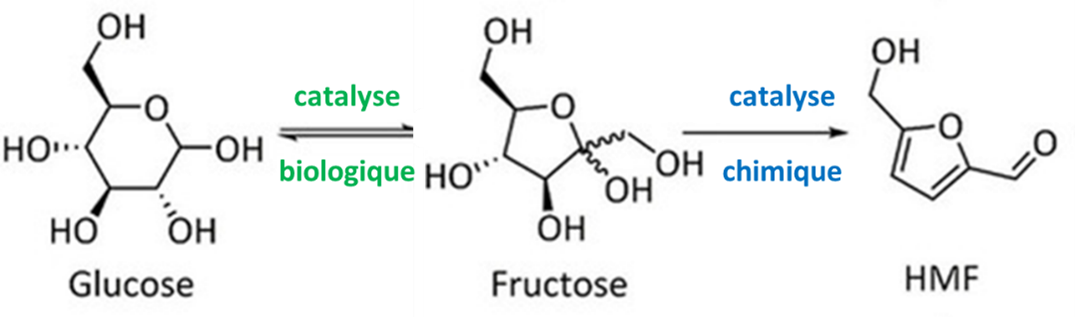
The isomerization of glucose into fructose is carried out very selectively2 by biological catalysis using an enzyme called glucose isomerase, with a final mixture of sugars made up of 55% fructose and 45% glucose. In order to increase the efficiency of this reaction by enabling the enzyme to maximize glucose conversion, it is necessary to consume the fructose produced throughout the process. Unfortunately, the acid chemical catalyst used to convert the fructose into HMF destroys the enzyme from the first step and it is not possible to have the two reactions take place in the same reactor.
2 i.e., without generating by-products
An innovative strategy for continuous fructose sampling
Researchers therefore came up with an innovative strategy using an H-shaped reactor, with two compartments and an ingenuous system for transporting the fructose from the first to the second [4]. This system uses a Y-B(OH)2 transporter to extract fructose from the first reactor, leaving the glucose behind, enabling the continued conversion of the latter by the enzyme.
To develop the process, numerous parameters were studied and optimized within the two compartments: pH, temperature, agitation speed, transporter quantity.
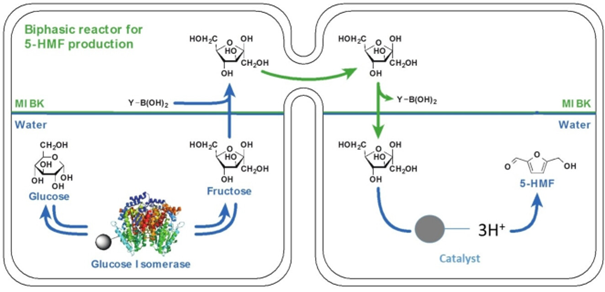
There are a number of advantages associated with this reactor:
- Absence of cohabitation and inhibition between biological and chemical catalysts;
- Separation of the two aqueous reaction phases by the organic transport phase;
- Increased glucose conversion by the enzyme;
- Facilitated 5-HMF recovery in the second reactor with minimal impurities.
In addition, the transporter is regenerated following fructose transport and returns to recover fructose thanks to an efficient system of blades and agitation.
Thanks to the new system developed [5], the yield from the first isomerization step was increased from 55 to 80% and an overall HMF yield of 31% was obtained without the need for an intermediate treatment step. This is a significant development in the field of hybrid catalysis and should pave the way for further such developments relating to the efficient conversion of renewable resources.
References :
[1] Thesis by Marie Guehl, « Nouveau concept de catalyse hybride pour la transformation de la biomasse », Science@IFPEn issue 31, December 2017,
[2] F. Dumeignil, M. Guehl, A. Gimbernat, , M. Capron, N. Lopes Ferreira, R. Froidevaux, J-S. Girardon, R. Wojcieszak, P. Dhulster, D. Delcroix, From sequential chemoenzymatic synthesis to integrated hybrid catalysis: taking the best of both worlds to open up the scope of possibilities for a sustainable future, Catal. Sci. Technol., 2018, 8, 5708-5734, https://doi.org/10.1039/C8CY01190G
[3] A. Gimbernat, M. Guehl, N. Lopes Ferreira, E. Heuson, P. Dhulster, M. Capron, F. Dumeignil, D. Delcroix, J-S. Girardon, R. Froidevaux, From a Sequential Chemo-Enzymatic Approach to a Continuous Process for HMF Production from Glucose, 2018, 8, 335-355, https://doi.org/10.3390/catal8080335
[4] A. Gimbernat, E. Heuson, F. Dumeignil, D. Delcroix, J-S. Girardon, Reactor development for a one-step hybrid catalytic conversion of D-glucose to HMF, ChemCatChem, 2024, 16, https://doi.org/10.1002/cctc.202300713
[5] IFPEN patent FR3077221
Scientific contacts : kim.larmier@ifpen.fr, nicolas.lopes-ferreira@ifpen.fr
You may also be interested in
Chimie biosourcée : de l’importance d’acquérir un grand nombre de données biologiques
Hybrid catalysis can better deal with bio-based substances
THÈSE DE MARIE GUEHL