Zeolites are microporous crystalline aluminosilicates that exist in the natural state and can also be synthesised for a wide range of applications, from the biomedical industry to the production of renewable energy. The introduction of germanium during the synthesis process has made it possible to obtain new zeolite-type solids, which have the advantage of pores that are often larger, giving them significant potential for use in catalysis. In fact, these large pores are accessible by molecules of a larger size, which opens up prospects for conversion of bulky molecules, such as those present in some petroleum fractions or in biomass.
However, these silicogermanates exhibit very low amounts of acid sites, while many reactions are catalysed by acid sites, and they are often unstable in aqueous state, which poses a barrier to their use. Post-treatment stages are therefore needed to acidify the silicogermanates and stabilise them.
A study by numerical simulation, using density functional theory (DFT), has initially demonstrated the thermodynamic feasibility of the stabilisation process [1]. This was then confirmed by experiment, thanks to the silicon enrichment of the zeolite framework, achieved by treatment in gas phase (with SiCl4) [2,3], before incorporation of aluminium in aqueous phase (with a compound known as PAC), which is designed to acidify the solid (figure a).
This work was carried out entirely at IFPEN as part of a doctoral thesis* and has resulted in a new stable zeolite, named IZM-7 [2,4], produced from silicogermanate IM-12. This solid contains virtually no remaining germanium, but retains its large pore size. The evaluation of its properties for catalysis was carried out in collaboration with Katholieke Universiteit Leuven. The results obtained demonstrate promising behaviour (figure b), in that the catalytic activity in the chosen model reaction is greater than that of conventional zeolites (USY). This work opens up potential avenues for the catalytic use of stable silicogermanate derivatives.
For her work on the paper, doctoral researcher Elsy El Hayek was awarded the 2022 Denise Barthomeuf Prize by the French Zeolites Group (GFZ).
Click on the picture to enlarge
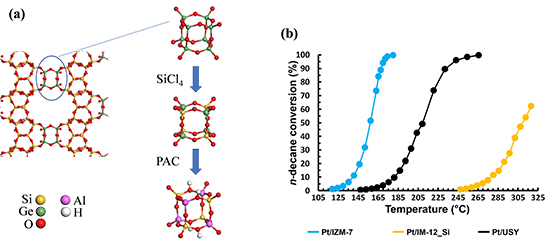
Figure (a): Structure of silicogermanate IM-12 and expected effect of the substitution strategy implemented.
Figure (b): Catalytic performances in hydroconversion of n-decane on bifunctional catalysts involving a conventional zeolite (USY), the IM-12 zeolite treated with SiCl4 (IM-12_Si) and the new aluminosilicate IZM-7 (the solid is all the more active given the low temperature required to achieve a given conversion).
* Thesis title: “New acid zeolites obtained from silicogermanates”, E. El Hayek, Claude Bernard Lyon 1 University, 2020
References:
- Ab initio investigation of the relative stability of silicogermanates and their (Alumino) Silicates counterparts, E. El Hayek, B. Harbuzaru, J. Martens, C. Chizallet, Microporous Mesoporous Mater. 306 (2020) 110425–110434.
>> DOI : 10.1016/j.micromeso.2020.110425
- IZM-7: A new stable aluminosilicogermanate with a promising catalytic activity, E. El Hayek, G. Vanbutsele, S. Radhakrishnan, M. Rivallan, E. Soyer, C. Bouchy, E. Breynaert, J. Martens, C. Chizallet, B. Harbuzaru, J. Catal.
>> DOI : 10.1016/j.jcat.2021.11.006
- Method for stabilisation of microporous silicogermanate in the presence of SiCl4, E. El Hayek, B. Harbuzaru, C. Chizallet, J. A. Martens, INPI Number : 3101788, publication date: 16 April 2021.
- UTL framework-type material with good structural resistance in the presence of water, a high microporous volume and active sites in its framework, E. El Hayek, B. Harbuzaru, C. Chizallet, J. A. Martens, INPI Number : 3106130, publication date: 16 July 2021.
Scientific contacts: Céline Chizallet, Bogdan Harbuzaru
You may also be interested in
Shedding light on the external surface of zeolites by quantum calculation
Reaction dynamics in zeolites under the quantum calculation spotlight
Zeolites are nanoporous solids widely used as acid catalysts for the conversion of hydrocarbon molecules. However, determining the rates of the elementary steps of reaction mechanisms...